Agriculture
Environment and Nitrogen Use Efficiency
Nitrogen is an essential nutrient for all forms of life and is of fundamental importance for the healthy growth of plants. However, when lost to the soil, water, and air, nitrogen in agriculture can lead to considerable environmental problems.
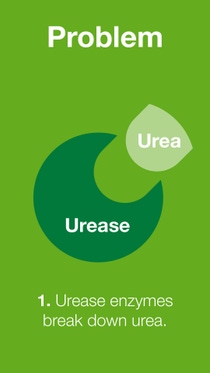
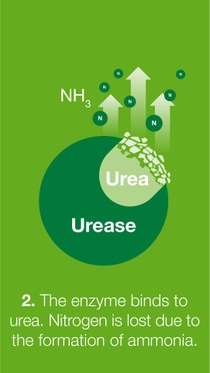
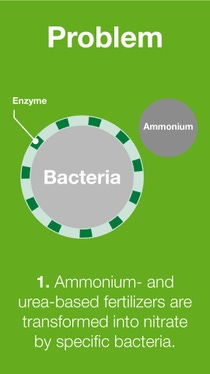
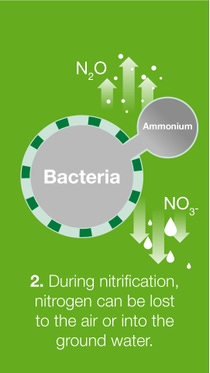
When fertilizer is transformed in the soil in various ways, several forms of nitrogen such as ammonia, nitrous oxide, nitrogen gas and/or nitrate are lost to the air or groundwater.
The excess of nutrients in the environment is a major source of air, soil, and water pollution, which negatively impacts biodiversity and climate. Directly related to nitrogen management, the EU Commission’s European Green Deal has set goals to reduce nutrient losses by at least 50% by 2030. BASF’s nitrogen stabilizer technologies are integral tools toward BASF Agricultural Solution’s sustainability commitment to reduce up to 30% CO2 emissions per ton of crop.
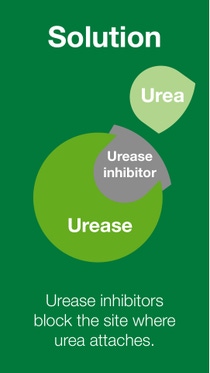
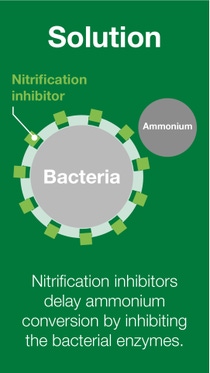
BASF’s nitrogen management portfolio of urease and nitrification inhibitors offers a solution for the reduction of nitrogen losses, and therefore, increasing nitrogen use efficiency.
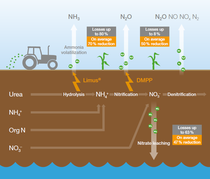
Ma et al. 2023
If you have questions or want more information, please send us an e-mail.
Related topics
Find out which of our nitrogen use efficiency products are available in your country.














.png?vid=_rmkx0OTb7pju1i9xE8Fjj8zoLogy3LN)